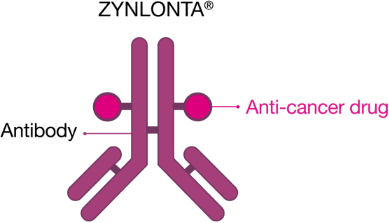
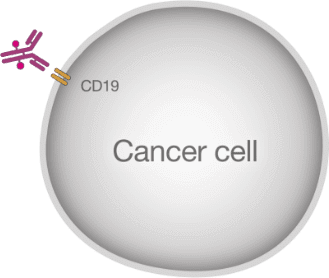
What is ZYNLONTA®?
ZYNLONTA® is a prescription medicine used to treat adults with certain types of large B-cell lymphoma that has come back (relapsed) or that did not respond to previous treatment (refractory), who have already received two or more treatments for their cancer.
It is not known if ZYNLONTA® is safe and effective in children.
The approval of ZYNLONTA® is based on a type of response rate. There is an ongoing study to confirm the clinical benefit of ZYNLONTA®.
IMPORTANT SAFETY INFORMATION
What are the possible side effects of ZYNLONTA®?
ZYNLONTA® may cause serious side effects, including:
- Fluid retention, including capillary leak syndrome (CLS). Fluid retention has happened in people treated with ZYNLONTA®, and can be serious and become life-threatening. Too much fluid buildup in your body can cause fluid to leak from blood vessels into the body’s tissues. This condition is called capillary leak syndrome (CLS). Tell your healthcare provider and get emergency help right away if you develop new or worsening signs and symptoms of fluid retention including CLS:
- swelling or puffiness
- rapid weight gain
- chest pain
- trouble breathing
- feeling unusually weak or tired
- dizziness or feeling faint especially when standing up
- Low blood cell counts (platelets, red blood cells, and white blood cells). Low blood cell counts are common with ZYNLONTA® but can also be serious or severe. Your healthcare provider will monitor your blood counts during treatment with ZYNLONTA®. Tell your healthcare provider right away if you get a fever of 100.4°F (38°C) or above, or any bruising or bleeding
- Infections. Serious infections, including infections that can cause death, have happened in people treated with ZYNLONTA®. Tell your healthcare provider right away if you get new or worsening signs or symptoms of infection
- Liver problems, including Drug-Induced Liver Injury (DILI). Liver problems have happened in people treated with ZYNLONTA®, and can be severe, life-threatening, or cause death. Your healthcare provider will do blood tests to monitor your liver function before and during treatment with ZYNLONTA®. Tell your healthcare provider if you get stomach-area (abdomen) discomfort, itching of the skin, dark or “tea-colored” urine, or yellowing of your skin or the whites of your eyes (jaundice)
- Read full Important Safety Information
- Skin reactions. Serious skin reactions have happened in people treated with ZYNLONTA®. Tell your healthcare provider if you get new or worsening skin reactions, including sensitivity to sunlight, skin rash, peeling, redness or irritation. You may burn more easily or get severe sunburns
Your healthcare provider may temporarily stop your treatment, lower your dose or permanently stop treatment with ZYNLONTA® if you have certain severe side effects.
The most common side effects of ZYNLONTA® include:
- feeling tired or weak
- skin rash
- swelling
- nausea
- muscle or joint pain
- increase in blood sugar (hyperglycemia)
- changes in certain blood or laboratory tests
ZYNLONTA® may cause fertility problems in males which may affect your ability to father children. Talk to your healthcare provider if this is a concern for you.
These are not all of the possible side effects of ZYNLONTA®.
What should I avoid while receiving ZYNLONTA®?
Avoid or limit your exposure to sunlight, including sunlight through glass, such as buildings or vehicle windows and artificial sunlight such as sunlamps or tanning beds. Exposure to sunlight during treatment with ZYNLONTA® can cause skin reaction or rash. Use sun protection measures such as sunscreen and wear loose-fitting clothes that cover your skin while out in sunlight.
Before you receive ZYNLONTA®, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines that you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get new medicine.
You may report side effects to the FDA at (800) FDA-1088 or www.fda.gov/medwatch. You may also report side effects to ADC Therapeutics at 1-855-690-0340.
Please see the full Prescribing Information for ZYNLONTA®, including Patient Information, for additional Important Safety Information.